LeBeau, Yule, Groblewski, Sneyd, 1999
Model Structure
The production of the intracellular signalling factor inositol 1,4,5-triphosphate (IP3) and the subsequent release of Ca2+ stored in intracellular organelles is a fundamental cellular signalling function. The inositol triphosphate receptor (IPR) is an IP3-activated Ca2+ channel in the ER. The properties of IP3-dependent intracellular calcium oscillations in pancreatic acinar cells depend on the agonist used to stimulate them.
This agonist-dependency is captured in Andrew P. LeBeau et al's 1999 mathematical model of agonist-specific intracellular calcium oscillations in pancreatic acinar cells. They assume that the complete IPR is composed of four functionally identical, independent subunits, (see below). IP3 must be bound to all four subunits for the receptor to be in the conducting state.
The complete original paper reference is cited below:
Agonist-dependent Phosphorylation of the Inositol 1,4,5-Triphosphate Receptor. A Possible Mechanism for Agonist-specific Calcium Oscillations in Pancreatic Acinar Cells, Andrew P. LeBeau, David I. Yule, Guy E. Groblewski and James Sneyd, 1999, The Journal Of General Physiology , 113, 851-871. (Full text and PDF versions of the article are available to subscribers of the JGP website.) PubMed ID: 10352035
The raw CellML description of the dynamic model of the type-2 inositol triphosphate receptor can be downloaded in various formats as described in .
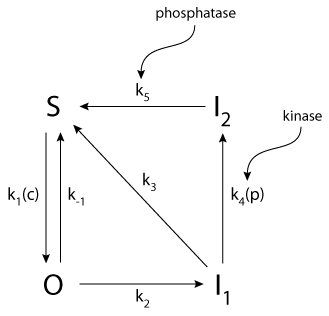 |
| A diagram of the receptor states of the model of the IP3 receptor. S denotes the fraction of the subunits in the shut state. Binding of IP3 causes the receptor to be converted to the open state O. O is a relatively unstable state and the subunits will progress through to the more stable I1 (inactivated) state in which IP3 is still bound but the channels do not conduct. I2 represents a second inactivated state of the receptor in which IP3 is no longer bound. |
