Ciliberto, Tyson, 2000
Model Status
This model has been edited to include initial concentrations and correct dimensional errors. The model runs in COR and PCEnv to produce oscillating graphs but has not been determined to exactly replicate the experimental results.
ValidateCellML verifies this model as valid CellML with consistent units.
Model Structure
During normal mitotic cell division, eukaryotic cells will replicate their DNA during the S-phase of the cell cycle, and then divide during the M-phase. S and M phases are temporally separated by gaps, called the G1 and G2 phases respectively. However, in early development, when the main aim of embryo is to increase cell number by a series of rapid cell divisions, the cell cycle has a number of unique features:
-
interdivision time is short and nearly constant;
-
S and M phases alternate in time without intervening G1 or G2 phases;
-
cell division is nearly synchronous and most cells take part;
-
and the nucleocytoplasmic ration (NCR) of cells increases dramatically.
It is believed that the NCR controls the cell cycle characteristics during the early stages of devlopment. This idea has been incorporated into the following mathematical model of early devlopmental cell cycle control.
The molecular components involved in mitotic control have been studied extensively in the last decade, and a control system, common to all eukaryotes, has been elucidated. This has been the subject of several mathematical models, including models which use budding yeast as the experimental organism:
-
Modelling the control of DNA replication in fission yeast, Novak and Tyson, 1997
-
Mathematical Model of the Fission Yeast Cell Cycle, Novak et al., 1998
-
Ciliberto et al. Modelling the Morphogenesis Checkpoint in the Budding Yeast Cell Cycle, 2003
and those which use the Xenopus oocyte and embryo: Ciliberto et al. A Kinetic Model of the Cyclin E/Cdk2 Developmental Timer in Xenopus laevis embryos, 2003.
In the Ciliberto and Tyson 2000 publication described here, the authors develop a mathematical model based on the molecular interactions underlying early embryonic cell cycle control (see the figure below). By introducing the NCP ratio into the molecular mechanism, through model simulations the authors were able to reproduce many of the physiological features of early sea urchin development. Since the cell cycle controls appear to be common to all eukaryotes, this model can also potentially be applied to other species.
The complete original paper reference is cited below:
Mathematical Model for Early Development of the Sae Urchin Embryo, Andrea Ciliberto and John J. Tyson, 2000, Bulletin of Mathematical Biology , 62, 37-59. (A PDF version of the article is available to subscribers on the Bulletin of Mathematical Biology website.) PubMed ID: 10824420
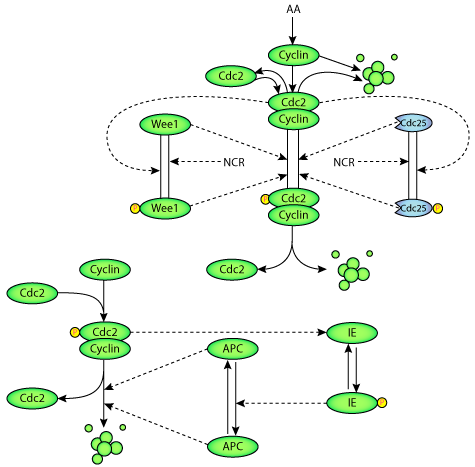 |
| The M phase control system of the cell cycle. The upper part of the figure shows the two positive feedback loops involving active MPF (Cdc2-cyclin complex) and inactive MPF (the phosphorylated Cdc2-cyclin complex). Cyclin is produced at a constant rate from a pool of amino acids (AA) and immediately binds to free Cdc2, producing active MPF. All forms of cyclin - free, active and inactive MPF - are degraded by the same process. The lower part of the figure shows that the degradation of cyclin is the result of a negative feedback loop involving a ubiquitin-ligation complex (APC) and a putative intermediary enzyme (IE). |
